“Nature builds the world with forms and numbers. “
Helena Bławatska
Fermions and bosons are the elementary molecules from which atomic nuclei are formed. Atomic nuclei are the central parts of atoms made up of protons, neutrons, and electrons. The sets of atoms, on the other hand, are made up of chemical elements. Chemical elements and their compounds in which they occur in nature or are obtained through the production process are called substances. A chemical reaction is usually required to make a substance. This is the process of converting one chemical element into another.
In this article you will learn:
- What atoms consist of and how they connect.
- About the evolution of chemical elements.
- And you will also learn about the relationship of elements with the ancient elements of nature.
According to the German philosopher Leibniz, a substance is also a monad, i.e. a simple substance included in complex things. They are atoms of nature, i.e. the soul in which the whole world is reflected. The word soul describes a combination of certain elements – nitrogen, hydrogen, and oxygen. I wrote about this in one of the previous articles.
Elements have specific chemical and physical properties and cannot be broken down into other substances by simple chemical reactions. They also create what we traditionally call matter in our reality, that is, everything that takes up space and has mass.
118 elements make up an infinite number of chemical compounds, of which only 92 occur naturally, including 4 elements common to all living organisms. These include oxygen, carbon, hydrogen, and nitrogen. These elements make up 96% of the human body and are the main components of DNA.
Elements make up physical bodies in three states of aggregation – solid, liquid, and gas. They are the counterparts of what we call spirit, soul, and physical body.
Chemical elements

Chemical elements
The smallest unit of matter which retains all the chemical properties of an element is the atom.
Everything that surrounds us is made of atoms. Atoms interact with other atoms or groups of atoms. Atoms combined into larger structures form molecules. A molecule of two atoms is a chemical compound. Atoms also tend to attract or donate electrons and bond with each other. These interactions between atoms, and their many combinations, make life possible.
Atoms are 99.99% made of a vacuum, which is a magnetic field.
Most of their mass is concentrated in the nucleus. An atomic nucleus is made up of charged particles called protons and neutral uncharged (electrically neutral) particles called neutrons. They are surrounded by a cloud of electrons, i.e. light negatively charged particles that circle them. The attraction between positively charged protons and negatively charged electrons keeps the atom in equilibrium. The protons, neutrons, and electrons of all elements are identical, but the atoms differ in their number. The number of protons in the nucleus determines the type of element and the properties – the number and configuration of electrons surrounding the nucleus.
Electrons are much lighter than protons, only about 1/1800 of the atomic mass, so they do not contribute much to the total atomic mass of the element. On the other hand, they greatly affect the charge of the atom since each electron has a negative charge equal to the positive charge of the proton. In uncharged, neutral atoms, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. The positive and negative charges cancel out, leading to the formation of an electrically neutral atom.
The electrons orbit the nucleus along a path strictly defined in terms of energy, which is called the stationary orbit.
In this state, they neither consume nor give up energy. However, they can change their orbit. It involves giving up and consuming energy in the form of portions called quanta. It can therefore be said that an atom consists of two areas a positively charged nucleus and a negatively charged electron sphere.
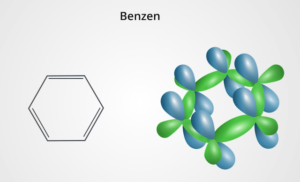
By combining, atomic orbitals form molecular orbitals, i.e. bonds. Strong or weak bonds with other atoms organize them into molecules or crystals. The shapes of the orbitals describing a given electron in an atom or molecule depend on the location of these electrons concerning the nuclei and other electrons. Both bond constants and weaker temporary bonds are essential to our body chemistry and the existence of life. These chemical bonds are nothing but the Vesica Piscis shape from Sacred Geometry. Look at the picture below, do the bonds that make up the chemical benzene resemble the flower of the life circle?
Atoms are trying to achieve the most stable, or lowest energetically, state they can, which simply means that they want to become solid – the heaviest element of uranium. They become stable when they follow the octet rule, i.e. when they have eight valence electrons – those placed in the last shell – orbit. If they don’t have such an arrangement, they try to achieve it by gaining or losing, or splitting electrons in chemical bonds. Why eight? This is due to the geometry of space represented in the Sacred Geometry by the Egg of Life. Atoms evolve from energy to matter, from the sun to earth and stars to planets.
In the process of uranium decay, helium is created, supplying the entire process of the formation of chemical elements with energy based on resonance. Uranus is at the core of the Earth and helium is at the core of the Sun.
The evolution of the elements was presented in 1888 by William Crooks in the form of a three-dimensional model – a spiral. The Crook’s spiral shows a swinging movement, drawing eights in space. It represents the mirror image of the motion of a sound wave, resulting in the formation of antimatter. He put hydrogen in the first place, but the first atom was helium because its atom has the perfect symmetry. Only the destruction of this symmetry results in evolution, and thus the formation of new chemical elements. In addition, the seers Annie Besant and Charles Webster Leadbeater, while examining its structure, noticed that there is a complete hydrogen atom inside it.
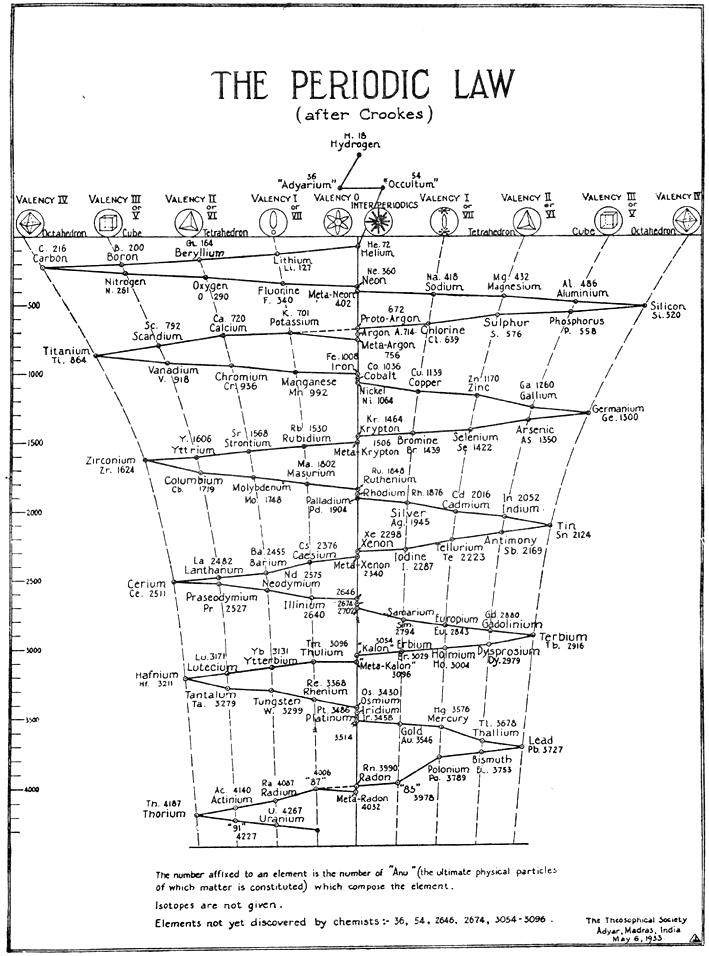
https://www.meta-synthesis.com/webbook/35_pt/pt_database.php?PT_id=646
Therefore, the helium element is the element of all life.
Helium (He-4) is the perfect symmetry of the universe as it has 2 electrons, 2 protons, and 2 neutrons. After the helium atom was saturated with energy (information), a hydrogen atom was precipitated, with 1 electron 1 proton, and 0 neutrons. It is therefore duality – asymmetry. From hydrogen, deuterium is formed – the isotope of hydrogen, which consists of 1 electron, 1 proton, and 1 neutron. Deuterium produces tritium, the unstable isotope of hydrogen, consisting of 1 electron, 1 proton, and 2 neutrons.
As a result of its decay, helium (He-3) is formed, consisting of 2 electrons, 2 protons, and 1 neutron. Then lithium (Li 6) is formed, consisting of 3 electrons, 3 protons, and 3 neutrons, then lithium (Lithium 7) – 3 electrons, 3 protons, and 4 neutrons. Next beryllium with the number of electrons 4, protons 4, and neutrons 5. Then boron (B 10), which consists of 5 electrons, 5 protons, and 5 neutrons, and boron (B 11), which consists of 5 electrons, 5 protons, and 6 neutrons.
Next, carbon (C12), which has 6 electrons, 6 protons, and 6 neutrons, and carbon (C13), which has 6 electrons, 6 protons, and 7 neutrons. Nitrogen (N 14) has 7 electrons, 7 protons, and 7 neutrons, and nitrogen (N 15) has 7 electrons, 7 protons, and 8 neutrons. Then oxygen (O16), has 8 electrons, 8 protons, and 8 neutrons, oxygen (O17), which has 8 electrons, 8 protons, and 9 neutrons, and oxygen (O18), which has 8 electrons, 8 protons, and 10 neutrons. Fluorine has 9 electrons, 9 protons, and 10 neutrons. Then neon (Ne-20) – an “improved” version of helium with 10 electrons, 10 protons, and 10 neutrons, (Ne-21) with 10 electrons, 10 protons, and 11 neutrons, (Ne-22) with 10 electrons, 10 protons, and 12 neutrons.
Neon is the beginning of a new cycle.
Each newly created element has more electrons, protons, and neutrons than the previous one, but it is the overload of neutrons that creates the new element. The neutron is the sound wave responsible for energizing the new level of consciousness in the understanding of the new “improved” information level, which is the number zero in vortex mathematics.
Bonds between atoms with an excess or a shortage of electrons – ions with opposite charges are ionic bonds. Atoms also, in addition to accepting and losing electrons, can share them, thus creating covalent bonds. These types of bonds are found, for example, in our DNA, water, or carbon dioxide molecules. The more electrons share the atoms, the stronger their bonds. These types of bonds can also be polarized. It means that electrons are unevenly divided and spend more time closer to one atom than the other. Then slightly positive and slightly negative, or non-polarized charges arise.
The electrons are in constant motion and their atoms or molecules may clump together at times, creating a partial negative charge in one part of the molecule and a partial positive charge in the other. If a molecule of this type is placed next to another, it can cause a similar redistribution of the charge in it. Then the temporary charges of one other molecule will attract each other. Such weak bonds between atoms are hydrogen bonds and London dispersion forces.
Orbitals are not something material, but a mathematical function that describes the probability of finding an electron.
“The mixing “of orbitals is the hybridization of the atom. It allows you to define the shape of the molecule. Two overlapping orbitals are a chemical bond. Depending on the number and type of orbitals involved in hybridization, various types can be distinguished, and the corresponding spatial arrangement of hybridized orbitals.
Shapes of orbitals:
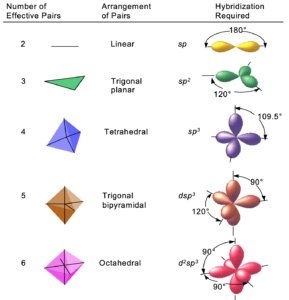
IB2 Chemistry at TASIS
Orbitals show the way electricity flows in the Universe, i.e. the flow of female electrostatic energy (electrons), and the flow of male electrostatic energy – positive ions (protons). Neutrons are the flow of kinesthetic energy – sound, and the vacuum in an atom is the magnetic field.
For centuries, mankind has associated the creation of the world with the 5 elements.
These elements were earth (carbon), water (hydrogen), air (oxygen), fire (helium), and ether (nitrogen) – the basic building blocks of the world. There were also added 3 additional elements mercury, sulfur, and salt, which symbolized the elementary qualities of metallicity, flammability, and solubility.
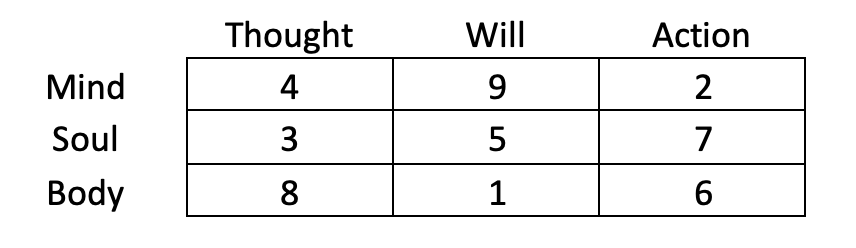
Similar elements can be found in the Chinese tradition of 5 transformations. The numbers of Marko Rodin’s mathematics that make up the Chinese Lo Shu square are related to 5 transformations – the elements, and according to the intuitive messages I received, each transformation corresponds to a different root:
- 2, 5, and 8 correspond to the earth’s element, hydrogen
- 7 and 6 metal element – oxygen
- 1 element of water – nitrogen
- 3 and 4 elements of wood – coal
- 9 element of fire – helium
Each number represents a direction in the space of the center, North, South, East, West, North-East, North-West, South-East, and South-West.
When we put these roots in place of numbers, this square looks like this:

And the interactions between these elements, when we replace them with elements, look like this:
- Coal kindles Hel (the earth kindles the sun)
- Helium forms Hydrogen,
- Hydrogen contains Oxygen,
- Oxygen carries Nitrogen
- Nitrogen feeds Carbon (see the nitrogen cycle in nature)
When we read these interactions in this way, we see how the universe came into being and how its various elements are related to each other. It then turns out that ancient cultures were not as stupid as we think, and although they did not use the same names for phenomena as we did, they knew how the universe worked.
Source:
Annie Besant, Charles Webster Leadbeater “Occult Chemistry. Investigations by clairvoyant magnification into the structure of the atoms of the periodic table and some compounds.” („Chemia okultystyczna. Badania struktury anatomów pierwiastków układu okresowego i niektórych związków chemicznych metoda jasnowidzenia w powiększeniu.”), Instytut Rozekruis Pers, Wieluń, 2019

